|
Standardized procedures are based on a dilution method 1-3 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of cephalothin powder. The MIC’s should be determined using a standardized procedure. 
These MIC’s provide estimates of the susceptibility of bacteria to antimicrobial compounds. Quantitative methods are used to determine antimicrobial minimal inhibitory concentrations (MIC’s). Penicillin-resistant Streptococcus pneumoniae is usually cross-resistant to beta-lactam antibiotics. It has no activity against Pseudomonas spp. It is not active against most strains of Enterobacter spp., Morganella morganii, and Proteus vulgaris. Note-Methicillin-resistant staphylococci and most strains of enterococci ( Enterococcus faecalis ) are resistant to cephalosporins, including cephalexin. Streptococcus pneumoniae (penicillin-susceptible strains) 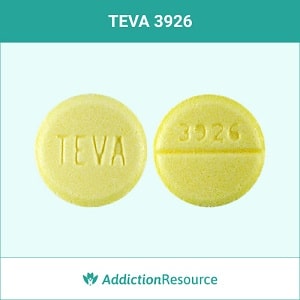
Staphylococcus aureus (including penicillinase-producing strains) Cephalexin has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section. In vitro tests demonstrate that the cephalosporins are bactericidal because of their inhibition of cell-wall synthesis. Inactive Ingredients: TABLETS: hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, and titanium dioxide. 
Inactive Ingredients: SUSPENSION: FD&C Red #40, cherry mixed fruit flavor (artificial flavors, benzyl alcohol, maltodextrin, and modified corn starch), silicon dioxide, sodium benzoate, sugar (fruit granulated), and xanthan gum.Įach tablet contains cephalexin monohydrate equivalent to 250 mg (720 μmol) or 500 mg (1,439 μmol) of cephalexin. In addition, the 250 mg capsule shell contains yellow iron oxide.Īfter mixing, each 5 mL of cephalexin for oral suspension USP will contain cephalexin monohydrate equivalent to 125 mg (360 μmol) or 250 mg (720 μmol) of cephalexin. Inactive Ingredients: CAPSULES: magnesium stearate, silicon dioxide, and sodium starch glycolate.Ĭapsule Shell and Print Constituents: black iron oxide, D&C Yellow #10 Aluminum Lake, FD&C Blue #1 Aluminum Lake, FD&C Blue #2 Aluminum Lake, FD&C Red #40 Aluminum Lake, gelatin, pharmaceutical glaze modified in SD-45, silicon dioxide or carboxymethylcellulose sodium, sodium lauryl sulfate, titanium dioxide and may contain propylene glycol. Cephalexin has a D-phenylglycyl group as substituent at the 7-amino position and an unsubstituted methyl group at the 3-position.Įach capsule contains cephalexin monohydrate equivalent to 250 mg (720 μmol) or 500 mg (1,439 μmol) of cephalexin. The cephalosporins differ from penicillins in the structure of the bicyclic ring system. Solubility in water is low at room temperature 1 or 2 mg/mL may be dissolved readily, but higher concentrations are obtained with increasing difficulty. It is a white crystalline solid having a bitter taste. The crystalline form of cephalexin which is available is a monohydrate. The isoelectric point of cephalexin in water is approximately 4.5 to 5. The compound is a zwitterion i.e., the molecule contains both a basic and an acidic group. The nucleus of cephalexin is related to that of other cephalosporin antibiotics. It is 7-(D-α-amino-α-phenylacetamido)-3-methyl-3-cephem-4-carboxylic acid, monohydrate.Ĭephalexin has the following structural formula: Caution should be exercised when cephalexin is administered to a nursing woman.Ĭephalexin, USP is a semisynthetic cephalosporin antibiotic intended for oral administration. The excretion of cephalexin in human milk increased up to 4 hours after a 500 mg dose the drug reached a maximum level of 4 mcg/mL, then decreased gradually, and had disappeared 8 hours after administration. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. There are, however, no adequate and well-controlled studies in pregnant women. Reproduction studies have been performed on mice and rats using oral doses of cephalexin monohydrate 0.6 and 1.5 times the maximum daily human dose (66 mg/kg/day) based upon mg/m2, and have revealed no harm to the fetus. 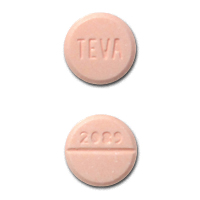
Reproduction studies have been performed on mice and rats using oral doses of cephalexin monohydrate 0.6 and 1.5 times the maximum daily human dose (66 mg/kg/day) based upon mg/m, and have revealed no harm to the fetus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
